THERE MAY BE A METHOD TO INCREASE LONGEVITY
EXCITING NEW DATA
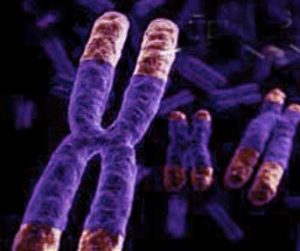
Many theories have been proposed to explain why people age. Most theories blame aging on damage to your DNA [deoxyribonucleic acid]. Before you can understand the theories regarding aging and how to increase longevity we must define our terms. The cells in your body contain your genetic information in the form of 23 pairs of chromosomes-each cell contains the same genetic information-the same 23 chromosomes-this is your DNA. Each chromosome is made up of sequences of 4 nucleic acids-adenine, guanine, thymine, and cytosine. You are a multicellular animal that requires cells [somatic cells] to divide in order for you to grow, maintain, and repair your body. The cells that produce eggs and sperm are not included in this discussion. During cell division-mitosis-one cell divides into 2 daughter cells each containing the same 23 pairs of chromosomes. However if this statement were completely true you could possibly live forever. During chromosome replication [mitosis] the enzymes [enzymes are chemicals that cause reactions] that duplicate the DNA cannot duplicate the DNA all the way to the end of the chromosome. Thus in every somatic cell division the end of the chromosome [the telomere] is shortened resulting in a small loss of DNA.
THE ENDS OF THE CHROMOSOMES ARE THE TELOMERES
The length of one type of cell telomere – your peripheral blood lymphocytes-can be easily measured by a simple blood test. The length of these telomeres is approximately 8,000 base pairs in newborns; approximately 3,000 base pairs in average adults; and down to 1,500 base pairs in the elderly. Remember we are measuring only the end of the chromosome since the entire chromosome has approximately 150 million base pairs. Each time a cell divides it loses between 30 to 200 base pairs. On the average a cell can divide 50 to 70 times before it dies. The cell death can be attributed to this loss of DNA-and theoretically this is the “aging process” [A.Ahmid and [J Am Geriatric Soc 49(8);1105-1109; 2001].Telomere length can be used as an objective marker for aging and longevity in a large homogeneous Amish population [OT Njajou et al; PNAS 104(29); 12135-12139]. In this setting telomere length [TL] is similar for men and women and similar for their children-thus a heritable link is present. TL does decrease with age.In a large biracial population TL was associated with more years of healthy life but not over all survival [O T Njajou et al; J Gerontology 64A(8); 2009; 860-864]. Thus relative telomere length is a marker for a healthy life style within a given individual.One path toward longer telomeres is through exercise. TL is preserved in healthy older adults who perform vigorous aerobic exercise and is positively related to maximal aerobic exercise capacity [TJ LaRocca et al; Mech Aging Develop 131(2); 2010; 165-167]. This may represent a novel molecular mechanism underlying the “anti-aging” effects of maintaining high aerobic fitness.In another study of study of 70 males and females ages 50 to 70 years moderate physical activity level over the past 5 years or longer was significantly associated with telomere length [AT Ludlow et al; Med Sci Sports Exerc 40(10}; 2008; 1764-1771]. Acute exercise may not be a strong enough stimulus to cause short-term changes to telomere length. The telomere length data from the Ludlow study indicates that a very low level of exercise and an extreme high level of exercise are associated with shorter telomere length compared with moderate levels of exercise over a long period of time.Telomeres are also particularly susceptible to free radicals that are thought to be major contributors to the aging process (Beckman and Ames 1998), and telomere shortening may provide a signal for knocking out cells with high levels of DNA damage (von Zglinicki 2002; di Fagagna et al. 2003). Once telomeres shorten to a critical length, a cell cycle checkpoint is triggered and cells enter a terminally nondividing state known as replicative senescence (Hornsby 2002). Telomeres can be elongated by the enzyme telomerase (Greider and Blackburn 1985), which is repressed in most normal adult somatic tissues because telomerase activation is necessary for cancer cells to maintain telomeres over multiple rounds of cell division (Forsyth et al. 2002). Thus, telomere shortening is thought to act as both an anticancer mechanism and contribute to the aging process (Campisi 2003; Hornsby 2006).It appears that individual TL during ones lifetime could provide a measure of that individual’s healthy lifestyle